detailed information§ Paraffin section immunofluorescence experiment steps
§ Dewaxing paraffin sections to water: Put the sections into xylene Ⅰ15min-xyleneⅡ15min-anhydrous ethanol Ⅰ5min-anhydrous ethanol Ⅱ5min-85% alcohol 5min-75% alcohol 5min-distilled water in order. § Antigen retrieval: Tissue sections are placed in a repair box filled with EDTA antigen retrieval buffer (PH8.0) for antigen retrieval in a microwave. 8 minutes in medium fire, 8 minutes in low fire, 7 minutes in low fire. During this process, the buffer solution should be prevented from over-evaporating, and never dry. After being allowed to cool, the slides were placed in PBS (PH7.4) and washed on a decolorizing shaker 3 times for 5 min each. (Repair fluid and repair conditions are determined according to the tissue) § BSA sealing: After the section is slightly dried, draw a circle around the tissue with a biochemical pen (to prevent the antibody from flowing away). Add 3% BSA in the circle to uniformly cover the tissue, and seal for 30 minutes at room temperature. § Add primary antibody: Gently shake off the blocking solution, drop PBS on the section and add the primary antibody prepared at a certain ratio, and place the section in a wet box and incubate overnight at 4 ° C. (Add a small amount of water to the wet box to prevent the antibody from evaporating) § Add secondary antibody: The slides are placed in PBS (PH7.4) and washed on the decolorization shaker 3 times for 5 min each. After the sections were slightly dried, the secondary antibody-covered tissues of the corresponding species of the primary antibody were added dropwise in the circle, and incubated at room temperature for 30 minutes in the dark. § DAPI counterstained nuclei: The slides were placed in PBS (PH7.4) and washed on a decolorization shaker 3 times for 5 min each. After the sections were slightly dried, DAPI staining solution was added dropwise in the circle, and incubated at room temperature for 10 minutes in the dark. § Coverslip: The slides were placed in PBS (PH7.4) and washed on a decolorizing shaker 3 times for 5 min each. The sections were slightly dried, and mounted with anti-fluorescence quenching mounts. § Microscopy: Take a slice and observe the image under a Nikon inverted fluorescence microscope. (Ultraviolet excitation wavelength 330-380nm, emission wavelength 420nm; FITC green excitation wavelength 465-495nm, emission wavelength 515-555 nm; CY3 red excitation wavelength 510-560, emission wavelength 590nm) § Interpretation of paraffin section immunofluorescence results The nucleus stained by DAPI is blue under ultraviolet excitation, and the positive expression is the corresponding red or green light labeled with fluorescein. 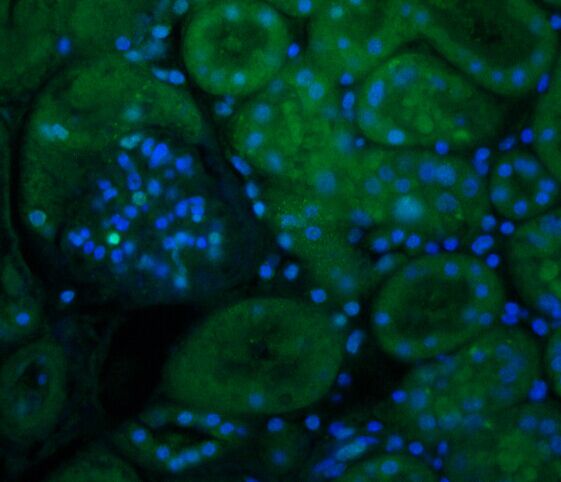 |
Contact Us
|

